Hypocalcemia
(Familial), Latent Tetany, and Calcification of Basal Ganglia Syndrome
- (Rare
clinical syndrome has features resembling those of
pseudohypoparathyroidism, pseudopseudohypoparathyroidism, and basal cell
nevus syndrome)
- Hypocalcemia is not
responsive to parathormone administration.
- Parathormone
administration produces a phosphate diuresis.
Hypocalcemia,
Neonatal
- Because
pH affects ionized calcium values, obtain free-flowing sample anaerobically
and seal in capillary tube until analysis
- Age 1-4 wks: serum
calcium <7 mg/dL or ionized calcium <4 mg/dL
- Age 1-2 days-associated
with
- Prematurity and low
birth weight occurs in ≤30% of infants
- Maternal diabetes found
in ≤25% of cases
- Birth asphyxia occurs in
≤30% of infants
P.607
- Age 5-10 days-associated
with
- Feeding of cow's milk
(increased serum phosphorus and decreased serum calcium)
- Rarely associated with
- Maternal hypercalcemia
or HP 313k1022d T
- Congenital absence of
parathyroid glands (see DiGeorge syndrome)
- Hypoproteinemia (e.g.,
nephrosis, liver disease)
- Maternal osteomalacia
- Renal disease (primary
rental tubular defect; decreased GFR causing phosphate retention)
- Iatrogenic disorders
(e.g., citrate administration during exchange transfusions)
- ○ When tetany syndrome is associated with a normal serum
calcium level or is not relieved by administration of calcium, rule out
decreased serum magnesium. Hypocalcemia associated with hypomagnesemia
does not respond unless hypomagnesemia is treated.
- ○ Serum phosphorus is >8 mg/dL when neonatal hypocalcemia is
due to high phosphate feeding. BUN is increased when neonatal hypocalcemia
is due to severe renal disease.
- Check serum calcium at
the following intervals:
- Infants of diabetic
mothers: 6, 12, 24, 48 hrs
- Infants with intrapartum
asphyxia: 1, 3, 6, 12 hrs
- Premature infants: 12,
24, 48 hrs
Hypocalciuric
Hypercalcemia, Familial (Familial Benign Hypercalcemia)
- (Rare
familial autosomal dominant disorder of chronic lifelong, asymptomatic,
nonprogressive, mild hypercalcemia due to resistance to action of
extracellular calcium on parathyroid gland and kidney; onset before age 10
yrs without renal stones, kidney damage, peptic ulcer; no response to
parathyroidectomy; parathyroid glands are histologically normal)
- Has many of same biochemical findings as primary HPT, including
- Mildly increased serum
total and ionized calcium.
- Inappropriately
increased (although within normal range) PTH level for hypercalcemia.
- Serum phosphorus is
slightly decreased or normal.
- Urinary cAMP is
increased in about one-third of patients.
- Urine calcium excretion is decreased or low normal despite hypercalcemia.
- ≤200 mg/24 hrs in
familial hypocalciuric hypercalcemia; calcium/creatinine clearance ratio
is usually <0.01 (but usually >0.02 in primary HPT).
- Up to 300 mg/24 hrs in
normal adult males.
- Increased (often >250
mg/24 hrs) in two-thirds of patients with HPT.
- Increased (often >500
mg/24 hrs) in patients with malignancies.
- Serum magnesium is mildly
increased in 50% of patients; this is the only condition in which serum
magnesium and calcium are both increased. Urine magnesium excretion is
decreased also.
- Renal function is
maintained with normal creatinine clearance.
- Serum 25-hydroxy-vitamin
D is normal and 1,25-dihydroxy-vitamin D is proportional to PTH level.
- Serum ALP is normal.
- No dysfunction of other
endocrine glands.
- May be family history of
hypercalcemia or failed parathyroidectomy attempts.
Hypoparathyroidism
- (Rare
disorder often detected in childhood; may be autoimmune disorder
associated with Addison's disease, diabetes mellitus, hypothyroidism, PA,
chronic hepatitis, moniliasis, malabsorption, or hypogonadism)
- See Table
13-8 and Fig. 13-6.
- Serum calcium is decreased (as low as 5 mg/dL) in presence of low or
inappropriately low PTH and normal serum magnesium, which affects PTH
secretion and action. Hypocalcemia stimulates PTH secretion in
pseudohypoparathyroidism but not in hypoparathyroidism.
P.608
- ○ Serum phosphorus
is increased (usually 5-6 mg/dL; as high as 12 mg/dL).
- Serum ALP is normal or
slightly decreased.
- Urine calcium is
decreased.
- Urine phosphorus is
decreased. Phosphate clearance is decreased.
- Serum PTH is decreased.
- Renal
resistance to PTH is shown by Ellsworth-Howard test
- PTH challenge (IV
administration of 200 IU of synthetic PTH) causes increased urine
phosphate (>10×) and cAMP in normal persons and in primary
hypoparathyroidism but little or no increase in urine phosphorus.
- Increased urine
phosphate (<2×) and cAMP in classical type I pseudohypoparathyroidism
or pseudopseudohypoparathyroidism.
- In type II
pseudohypoparathyroidism cAMP increases without phosphaturia.
- Decreased response may
occur in basal cell nevus syndrome.
- Alkalosis is present.
- Serum uric acid is
increased.
- OGTT results are flat
(due to poor absorption).
- CSF is normal, even with
mental or emotional symptoms or with calcification of basal ganglia.
- ○
Hypoparathyroidism should be ruled out in presence of mental and emotional
changes, cataracts, faulty dentition in children, associated changes in
skin and nails (e.g., moniliasis is frequent). One-third of these patients
may present as "epileptics."
- Congenital absence may be
associated with thymic aplasia (DiGeorge's syndrome).
Hypophosphatasia
- (Rare
[1 in 100,000 live births] autosomal recessive disease of bone
mineralization with radiographic changes and at least three different
clinical syndromes found in infants [most severe], children, and adults
[least severe])
- Serum ALP is decreased to ~25% of normal (may vary from 0 ≤
40% of
normal); is not correlated with severity of disease. Due to bone and
sometimes liver isoenzymes; normal ALP in intestine and placenta. Is
decreased in heterozygotes but the level cannot distinguish patients from
carriers.
- Serum calcium may be
increased in severe cases in newborns.
- Serum phosphorus is
normal.
- Serum and urine levels of phosphoethanolamine are increased (may be increased in
asymptomatic heterozygotes and useful for detection).
- Treatment with corticosteroids usually causes an increase in serum ALP (but it never
attains normal level) with a marked fall in serum calcium;
phosphoethanolamine excretion in urine continues to be high.
- Prenatal diagnosis by measurement of ALP in cultured
amniocytes, but activity in amniotic fluid is unreliable.
- Urine hydroxyproline is
low; in contrast, it is high in vitamin D-resistant rickets or
hyperphosphatasia.
Hypophosphatemia,
Primary
- (Familial
but occasionally sporadic condition of intrinsic renal tubular defect in
phosphate resorption)
- Serum phosphorus is always decreased in the untreated patient.
- Serum calcium is usually
normal.
- Serum ALP is often
increased.
- Bone biopsy shows a characteristic pattern of demineralization around osteocyte
lacunae.
Milk-Alkali
(Burnett's) Syndrome
- Increased serum calcium
(without hypercalciuria)
- Increased serum
phosphorus
- Mild alkalosis
P.609
- ○ The previous
section should suggest the diagnosis in a patient with peptic ulcer.
- Normal serum ALP
- Renal insufficiency with
azotemia (increased BUN)
- Metastatic calcinosis
Pseudohypoparathyroidism
- (Heterogeneous
group of inherited disorders with renal resistance to PTH action. Patients
may be short, stocky with round face, short metacarpals and metatarsals,
calvarial thickening, mental retardation.)
- Serum calcium, phosphorus, and ALP are the same as in hypoparathyroidism but
cannot be corrected by (or respond poorly to) administration of PTH (see
description of Ellsworth-Howard test,).
- Serum PTH level is normal or elevated.
- Renal resistance to PTH is shown by Ellsworth-Howard test (see previous section).
Pseudopseudohypoparathyroidism
- (Clinical
anomalies are the same as in pseudohypoparathyroidism.)
- Serum and urine calcium,
phosphorus, and ALP are normal.
- Ellsworth-Howard test
(see Hypoparathyroidism).
Pseudohypophosphatasia
- (Clinical
syndrome resembling hypophosphatasia)
- Serum ALP is normal.
Tetany
With Decreased Tissue Calcium
- Tetany associated with normal serum calcium, magnesium, potassium, and
carbon dioxide, responds to vitamin D therapy.
- Special radioactive calcium studies show decreased tissue calcium pool that returns toward
normal with therapy.
Tetany
Syndrome Due To Magnesium Deficiency
- Serum magnesium is decreased (usually <1 mEq/L).
- Serum calcium is normal
(slightly decreased in some patients)
- Blood pH is normal.
- Tetany responds to administration of magnesium but not of calcium.
Tests
for Diagnosis of Diabetes Mellitus and Hypoglycemia
C-Peptide,
Serum
- C-peptide is formed
during conversion of proinsulin to insulin; C-peptide serum levels
correlate with insulin levels in blood, except in patients with islet cell
tumors and possibly in obese patients.
Use
- Estimation of insulin
levels in the presence of antibodies to exogenous insulin.
- Diagnosis of factitious
hypoglycemia due to surreptitious administration of insulin, in which high
serum insulin levels occur with low C-peptide levels.
Increased
In
- Insulinoma
- Type II diabetes mellitus
P.610
Decreased
In
- Exogenous insulin
administration (e.g., factitious hypoglycemia)
- Type I diabetes mellitus
Fructosamine,
Serum
- Measures concentration of
nonlabile glycated serum proteins, giving a reliable estimate of mean
blood glucose levels during preceding 1-3 wks.
- Should primarily be
compared with previous values in same patient rather than with reference
range.
- Reference range in
nondiabetic persons: fructosamine = 2.4-3.4 mmol/L; fructosamine:albumin
ratio = 54-86 µmol/gm.
Use
To monitor treatment of diabetic patients
Interpretation
Correlates with HbA1c but is not affected by abnormal
hemoglobins, HbF, or increased RBC turnover and shows changed glucose levels
earlier; is cheaper, faster, less subjective than HbA1c
Interferences
- Changes in fructosamine
values correlate with significant changes in serum albumin or protein
concentrations. Abnormal values also occur during abnormal protein
turnover (e.g., thyroid disease) even though patients are normoglycemic.
Obviated by using fructose/albumin ratio.
- Dysproteinemias
- Increased serum bilirubin
may interfere.
- Possibly uremia, lipemia,
hemolysis, ascorbate.
Glycohemoglobin
(Glycated Hemoglobin)
- May be reported as HbA1c or as total of A1b, A1a, A1c
- Values may not be
comparable using different methodologies and even from different
laboratories using same methodology.
- Glucose combines with Hb
continuously and nearly irreversibly during life span of RBC (120 days);
thus glycated Hb is proportional to mean plasma glucose level during previous
6-12 wks.
- Glycated Hb predicts risk
of progression of diabetic complications.
- Glycosylated albumin
(half life ~14 days) has been used for monitoring degree of hyperglycemia
during previous 1-2 wks when glycated Hb cannot be used. Not yet shown to
be related to risk of progression of diabetic complications.
Use
- Monitor
diabetic patients' compliance with therapeutic regimen and long-term blood
glucose level control
- In known diabetics:
- 7% indicates good
diabetic control.
- 10% indicates fair
diabetic control.
- 13-20% indicates poor
diabetic control.
- When mean annual HbA1c
is <1.1× ULN, renal and retinal complications are rare, but
complications occur in >70% of cases when HbA1c is >1.7 ULN.
- Not presently recommended
for diagnosis of diabetes mellitus although ~85% sensitivity and
specificity each for screening.
Interpretation
- Dietary preparation or
fasting not required.
- Low sensitivity but high
specificity compared to OGTT, which has high sensitivity but low
specificity in diagnosis of diabetes mellitus.
P.611
- Increase almost certainly means diabetes mellitus if other factors (see below) are
absent (>3 SD above the mean has 99% specificity and ~48% sensitivity),
but a normal value does not rule out impaired glucose tolerance. Values
less than normal mean are not seen in untreated diabetes.
- May rise within 1 wk
after rise in blood glucose when therapy is stopped but may not fall for
2-4 wks after blood glucose decrease when therapy is resumed.
- Mean blood glucose in
first 30 days (days 0-30) before sampling glycated Hb contributes ~50% to
final glycated Hb value, whereas mean blood glucose in days 90-120
contributes only ~10%. Time to reach a new steady state is 30-35 days.
- When fasting blood
glucose = <110 mg/dL, HbA1c is normal in >96% of
cases.2
- When fasting blood
glucose = 110-125 mg/dL, HbA1c is normal in >80% of
cases.2
- When fasting blood
glucose = >126 mg/dL, HbA1c is normal in >60% of
cases.2
Normal (A1a, A1b, A1c
- For level of 4-20%, this
formula may estimate daily average plasma glucose: Mean daily plasma
glucose (mg/dL) = 10 × (glycohemoglobin level + 4)
- 1% increase in
glycohemoglobin is related to ~30 mg/dL increase in glucose.
Increased
In
- HbF above normal or 0.5%
(e.g., heterozygous or homozygous persistence of HbF, fetomaternal
transfusion during pregnancy)
- Chronic renal failure
with or without hemodialysis
- Iron-deficiency anemia
- Splenectomy
- Increased serum
triglycerides
- Alcohol
- Lead and opiate toxicity
- Salicylate treatment
Decreased
In
- Shortened RBC life span
- Presence of HbS, HbC,
HbD
- Hemolytic anemias (e.g.,
congenital spherocytosis)
- Acute or chronic blood
loss
- After transfusions
- Pregnancy
- Ingestion of large
amounts (>1 gm/day) of vitamin C or E
Insulin,
Plasma
Use
- Diagnosis of insulinoma
- Not clinically useful for
diagnosis of diabetes mellitus
Increased
In
- Insulinoma. Fasting blood
insulin level >50 µU/mL in presence of low or normal blood glucose
level. IV tolbutamide or administration of leucine causes rapid rise of
blood insulin to very high levels within a few minutes with rapid return
to normal.
- Factitious hypoglycemia
- Insulin autoimmune
syndrome
- Untreated obese patients
with mild diabetes. The fasting level is often increased.
- Patients with acromegaly
(especially with active disease) after ingestion of glucose
- Reactive hypoglycemia after
glucose ingestion, particularly when diabetic type of glucose tolerance
curve is present
P.612
Absent
In
Severe diabetes mellitus with ketosis and
weight loss. In less severe cases, insulin is frequently present but only at
lower glucose concentrations.
Normal In
- Hypoglycemia associated
with nonpancreatic tumors
- Idiopathic hypoglycemia
of childhood, except after administration of leucine
Insulin/C-Peptide
Ratio
Use
To differentiate insulinoma from factitious
hypoglycemia due to insulin
Interpretation
- <1.0 in molarity units
(or >47.17 µg/ng in conventional units)
- Increased endogenous
insulin secretion (e.g., insulinoma, sulfonylurea administration)
- Renal failure
- >1.0 in molarity units
(or <47.17 µg/ng in conventional units)
- Exogenous insulin
administration
- Cirrhosis
Proinsulin
Proinsulin level is normally ≤20% of
total insulin. Proinsulin is included in the immunoassay of total insulin, and
separation requires special technique.
Increased
In
- Insulinoma. Proinsulin
>30% of serum insulin after overnight fast suggests insulinoma.
- Factitious hypoglycemia
due to sulfonylurea use (see Table 13-14).
- Familial
hyperproinsulinemia-heterozygous mutation affecting cleavage of
proinsulin, leading to secretion of excess amounts of proinsulin.
- Non-insulin dependent
diabetes mellitus
Interferences
May also be increased in renal disease.
Tolerance
Test, Insulin
Administer 0.1 U insulin/kg body weight IV.
Use smaller dose if hypopituitarism is suspected. Always
keep IV glucose available to prevent severe reaction.
Normal
Blood glucose falls to 50% of fasting level
within 20-30 mins; returns to fasting level within 90-120 mins.
Increased
Tolerance In
- Blood glucose falls
<25% and returns rapidly to fasting level.
- Hypothyroidism
- Acromegaly
- Cushing's syndrome
- Diabetes mellitus (some
patients; especially older, obese ones)
Decreased
Tolerance In
- Increased sensitivity to
insulin (excessive fall of blood glucose)
- Hypoglycemic
irresponsiveness (lack of response by glycogenolysis)
- Pancreatic islet cell
tumor
P.613
|
|
|
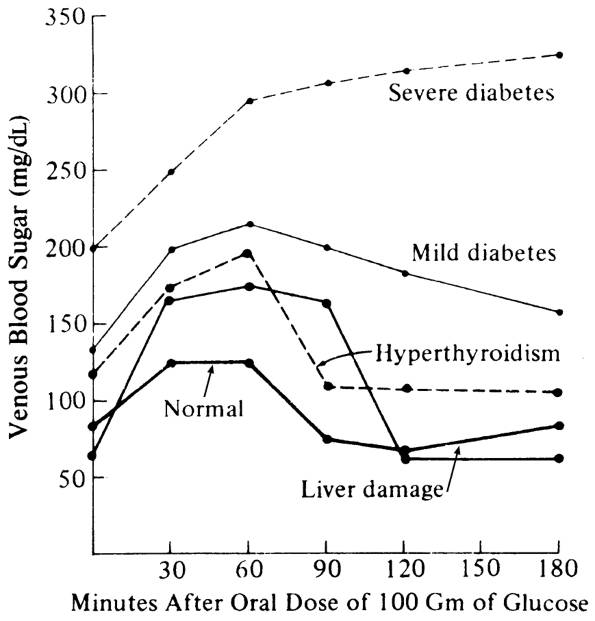
Fig. 13-8. Sample oral glucose tolerance
test curves in various conditions.
|
- Adrenocortical
insufficiency
- Adrenocortical
insufficiency secondary to hypopituitarism
- Hypothyroidism
- von Gierke's disease
(some patients)
- Starvation (depletion of
liver glycogen)
Tolerance
Test, Insulin Glucose
- Administer simultaneously
0.1 U insulin/kg body weight IV and 0.8 gm glucose/kg body weight orally.
- Insulin-sensitive
diabetics show little change in blood sugar.
- Insulin-resistant
diabetics show a diabetic glucose tolerance curve.
- Other changes parallel
those in the insulin tolerance test.
Tolerance
Test, Oral Glucose (Ogtt)
- See Fig.
13-8.
- Standards for OGTT: Prior
diet of >150 gm of carbohydrate daily, no alcohol, and unrestricted
activity for 3 days before test. Test in morning after 10-16 hrs of
fasting. No medication, smoking, or exercise (remain seated) during test.
Not to be done during recovery from acute illness, emotional stress,
surgery, trauma, pregnancy, inactivity due to chronic illness; therefore
is of limited or no value in hospitalized patients. Certain drugs should
be stopped several weeks before the test (e.g., oral diuretics, oral
contraceptives, phenytoin). Loading dose of glucose consumed within 5
mins: for adults = 75 gm, for children = 1.75 gm/kg (of ideal body weight
in obese children but never >75 gm), for pregnant women = 100 gm. Draw
blood at fasting, 30, 60, 90, 120 mins; 30-min sample offers little
additional information but can confirm adequate gastric absorption when
patient is nauseous.
P.614
Use
- OGTT should be reserved
principally for patients with "borderline" fasting plasma glucose levels
(i.e., fasting range 110-140 mg/dL).
- All pregnant women should
be tested for gestational diabetes with a 50-gm dose at 24-28 wks of
pregnancy; if result is abnormal, OGTT should be performed after
pregnancy.
- OGTT is not indicated in
- Persistent fasting
hyperglycemia (>140 mg/dL).
- Persistent fasting
normoglycemia (<110 mg/dL).
- Patients with typical
clinical findings of diabetes mellitus and random plasma glucose >200
mg/dL.
- Suspected gestational
diabetes.
- Secondary diabetes
(e.g., genetic hyperglycemic syndromes, after administration of certain
hormones).
- Should never be
performed to evaluate reactive hypoglycemia.
- Test is of limited value
for diagnosis of diabetes mellitus in children and is rarely indicated
for that purpose.
Interpretation
For diagnosis of diabetes mellitus in
nonpregnant adults, at least two values of OGTT should be increased (or fasting
serum glucose ≥140 mg/dL on more than one occasion) and other causes of
transient glucose intolerance must be ruled out. See Classification
of Diabetes Mellitus and Other Hyperglycemic Disorders,
Diabetes Mellitus, Gestational.
Decreased
Tolerance In
- Excessive peak
- Increased absorption
(normal IV GTT curve) with normal return to fasting level
- Mechanical causes (e.g.,
gastrectomy, gastroenterostomy)
- Hyperthyroidism
- Excess intake of glucose
- Decreased utilization
with slow fall to fasting level
- Diabetes mellitus
- Hyperlipidemia, types
III, IV, V
- Hemochromatosis
- Steroid effect
(Cushing's disease, administration of ACTH or steroids)
- CNS lesions
- Decreased formation of
glycogen with low fasting levels and subsequent hypoglycemia
- von Gierke's disease
- Severe liver damage
- Hyperthyroidism (normal
return to fasting level)
- Increased epinephrine
(stress, pheochromocytoma) (normal return to fasting level)
- Pregnancy (normal return
to fasting level)
- Drugs that may cause
increased serum glucose and/or impaired glucose tolerance
- Hormones (e.g., oral
contraceptives, thyroid hormone, ACTH or steroids, progestins)
- Antiinflammatory agents
(e.g., indomethacin)
- Diuretic and
antihypertensive drugs (e.g., thiazides, furosemide, clonidine)
- Neuroactive drugs (e.g.,
phenothiazines, tricyclics, lithium carbonate, haloperidol, adrenergic
agonists)
- Others (e.g., isoniazid,
heparin, cimetidine, nicotinic acid)
Increased
Tolerance In
- Flat peak
- Pancreatic islet cell
hyperplasia or tumor
- Poor absorption from GI
tract (normal IV GTT curve)
- Intestinal diseases
(e.g., steatorrhea, sprue, celiac disease, Whipple's disease)
- Hypothyroidism
- Addison's disease
- Hypoparathyroidism
- Late hypoglycemia
- Pancreatic islet cell
hyperplasia or tumor
P.615
- Hypopituitarism
- Liver disease
- See Glucose,
for effect of drugs.
- Difficulty in
interpretation has caused abandonment of other GTTs, such as IV GTT,
cortisone GTT.
Tolerance
Test, Tolbutamide
Administer 1 gm sodium tolbutamide IV
within 2 mins. Always keep IV glucose available to prevent
severe reaction.
Use
Most useful for diagnosis of insulinoma and
to rule out functional hyperinsulinism.
Interpretation
- In healthy persons,
glucose is a more potent stimulus for insulin release than tolbutamide,
but the opposite is true in insulinoma, which shows an exaggerated early
insulin peak (3-5 mins after injection) with a sustained elevation of
insulin and depression of glucose at 150 mins.
- The fall in blood sugar
is usually more marked in insulinoma than in functional hypoglycemia; more
importantly, the blood sugar fails to recover even after 2-3 hrs. A mean
serum glucose at 120, 150, and 180 mins after tolbutamide of ≤55
mg/dL in lean patients and 62 mg/dL in obese patients has a 95%
specificity and >95% sensitivity for insulinoma; this is the most
useful test.4 Other calculations of glucose and
insulin levels are less useful.
- In functional
hypoglycemia, return of blood sugar to normal is usually complete by 90
mins.
- Adrenal
insufficiency-normal or low curve
- Severe liver disease-low
curve
Diabetes
Mellitus and Other Hyperglycemic and Hypoglycemic Disorders
Beckwith-Wiedemann
Syndrome
- (Inherited
syndrome characterized by various abnormalities [e.g., macroglossia,
umbilical hernia, gigantism that may be unilateral, abnormal ear lobe
grooves, microcephaly])
- Symptomatic hypoglycemia
in ≤ 50% of patients, usually within first day, but may occur up to
3 days later; may be severe, difficult to control, and lasts for several
months.
- Hypocalcemia may occur.
- Polycythemia may occur.
- Cytogenetic studies are
normal.
Classification
of Diabetes Mellitus and Other Hyperglycemic Disorders
- Type I: immune-mediated
beta cell destruction diabetes mellitus (formerly called
insulin-dependent, juvenile-onset, ketosis-prone, or brittle diabetes
mellitus); represents 10-20% of diabetic patients. Autoantibodies are
present in 85-90% of cases. Insulin secretion is virtually absent. Plasma
C-peptide low or undetectable. Other autoimmune disorders may be present
(e.g., Graves' disease, Hashimoto's thyroiditis, Addison's disease, PA).
No autoantibodies in 10-15% of cases; strongly inherited.
P.616
- Type 2 (formerly called
non-insulin-dependent or adult-onset diabetes mellitus); represents 80-90%
of diabetic patients. Varies from predominantly insulin resistance with
relative deficiency to predominantly insulin secretory defect with insulin
resistance. Relative rather than absolute insulin deficiency. Not due to
autoimmunity or other disorders listed below. Plasma insulin may be normal
or increased but expected to be higher relative to blood glucose concentration.
Ketosis occurs with stress (e.g., infection) but seldom spontaneously.
Associated with dyslipidemia, obesity, increasing age, hypertension,
family history.
- Other specific types,
e.g.,
- Genetic defects of beta
cell function (e.g., chromosome 12, 7, 20). Formerly referred to as
maturity-onset diabetes of the young. Onset of mild hyperglycemia,
usually before age 25 yrs, and impaired insulin secretion. Autosomal
dominant inheritance.
- Genetic defects in
insulin resistance (e.g., leprechaunism, type A insulin resistance,
Rabson-Mendenhall syndrome, lipoatrophic diabetes)
- Diseases of exocrine
pancreas (e.g., pancreatitis, pancreatectomy, neoplasia, cystic fibrosis,
hemochromatosis)
- Endocrine disorders
(e.g., Cushing's syndrome, acromegaly, pheochromocytoma, aldosteronoma,
hyperthyroidism, glucagonoma)
- Drug/chemical induced
(e.g., glucocorticoids, phenytoin, beta-adrenergic agonists, pentamidine,
thiazides, interferon-alpha)
- Infections (e.g., CMV
infection, congenital rubella)
- Uncommon forms of
immune-mediated diabetes (e.g., anti-insulin receptor antibodies,
stiff-man syndrome)
- Other genetic syndromes
that may be associated with diabetes mellitus (e.g., Down syndrome,
Klinefelter's syndrome, Turner's syndrome, Friedreich's ataxia,
Huntington's chorea, Laurence-Moon-Biedl syndrome, porphyria,
Prader-Willi syndrome)
- Gestational diabetes
mellitus (see below)
Criteria for Diagnosis
- Diabetes Mellitus
- Random glucose >200
mg/dL when classical symptoms are seen or
- Fasting (>8 hrs)
serum glucose ≥126 mg/dL or
- 2-hr glucose >200
mg/dL after 75-gm glucose load. OGTT not recommended for routine use.
- Must be confirmed on
another day by any of the previous tests.
- Fasting blood glucose
≥126 mg/dL = provisional diagnosis of diabetes mellitus; must be
confirmed as noted previously.
- Diagnosis of "acute
metabolic decompensation with hyperglycemia" need not be confirmed on a subsequent
day.
- Impaired Glucose
Tolerance
- Fasting glucose
≥110 mg/dL but <126 mg/dL in nonpregnant adult.
- With OGTT, 2-hr value
≥140 and <200 mg/dL. Replaces terms latent
and chemical diabetes.
- Impaired Fasting Glucose
- Fasting glucose
≥110 mg/dL but <126 mg/dL or
- With OGTT, 2-hr value
≥140 but <200 mg/dL.
- In absence of pregnancy,
impaired glucose tolerance and impaired fasting glucose are risk factors
for future diabetes mellitus and cardiovascular disease; not clinical
entities.
- Other
causes of transient glucose intolerance must be ruled out before an
unequivocal diagnosis of diabetes mellitus is made
- Test asymptomatic
undiagnosed individuals every 3 yrs over age 45.
- Test at younger age if
- HDL cholesterol is
≤35 mg/dL or triglyceride is ≥250 mg/dL.
- Previous impaired
glucose tolerance or impaired fasting glucose.
- Obese.
- Has first-degree
relative with diabetes mellitus.
- Member of high-risk
ethnic population (e.g., black, Native American, Hispanic).
- Delivered baby weighing
>9 pounds.
- See sections on diabetic
nephrosclerosis, papillary necrosis, GU tract infection, serum
lipoproteins, etc.
P.617
|
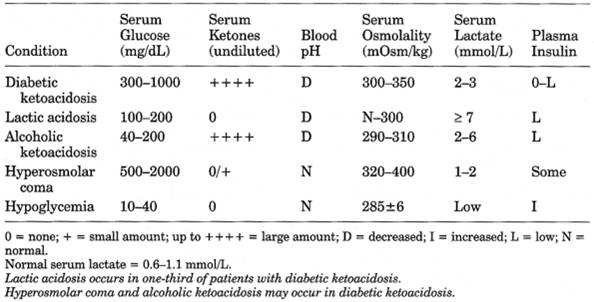
|
|
Table 13-11. Differential Diagnosis of
Diabetic Coma
|
Coma,
Nonketotic, Hyperosmolar Hyperglycemic
- (Due
to combination of severe dehydration caused by inadequate fluid intake and
insulin deficiency; occurs predominantly in type II diabetes mellitus)
- See Table
13-11 and .
- Blood glucose is very high, often 600-2000 mg/dL, but contrary to expectation in diabetic
coma, acidosis and ketosis are minimal and plasma acetone is not found.
- Serum osmolality is very high (normal = 280-300 mOsm/L). In mildly drowsy patients,
mean is 320 mOsm/L. At level of 350 mOsm/L, some confusion or some stupor
is seen. At level >350 mOsm/L, many patients are in coma. At 400
mOsm/L, most patients are obtunded. State of consciousness does not
correlate with height of acidemia.
- ○ Serum sodium may
be increased, normal, or decreased but is disproportionately decreased for
degree of dehydration due to marked hyperglycemia (artifactual decrease
1.6 mEq/L for every 100 mg/dL increase of serum glucose).
- Increased sodium with
marked hyperglycemia indicates severe dehydration.
- Serum potassium may be
increased (due to hyperosmolality), low (due to osmotic diuresis with
urinary loss), or normal depending on balance of factors.
- BUN is increased (70-90
mg/dL) more than in diabetic ketoacidosis.
- Laboratory findings due
to complications or precipitating factors
- Renal insufficiency in
90% of cases
- Infection (e.g.,
pneumonia)
- Drugs (e.g., steroids,
phenytoin, potassium-wasting diuretics such as thiazides and furosemide,
others [propranolol, diazoxide, azathioprine])
- Other medical conditions
(e.g., cerebrovascular or cardiovascular accident, subdural hematoma,
severe burns, acute pancreatitis, thyrotoxicosis, Cushing's syndrome)
- Glucose overloading or
use of concentrated glucose solutions (e.g., hyperalimentation, dialysis,
IV infusions in treatment of burns)
- Spontaneous in 5-7% of
cases
- Preexisting mild
diabetes mellitus type II
- Dehydration
- Clinical picture: A
middle-aged or older person with diabetes of recent onset or unrecognized
diabetes, who shows neurologic symptoms (e.g., convulsions or hemiplegia)
and then becomes stuporous or comatose
Diabetes
Mellitus, Gestational
- Hyperglycemia that
develops for the first time during pregnancy; affects ~4% of pregnant
women; most have return to normal glucose tolerance after delivery. 60%
become diabetic in next 16 yrs.
P.618
|

|
|
Table 13-12. Comparison of Diabetic
Ketoacidosis and Hyperosmolar Hyperglycemic Nonketotic Coma
|
P.619
- Diagnosis is necessary
for short-term identification of increased risk of fetal morbidity
(stillbirth, macrosomia, birth trauma, hypoglycemia, hyperbilirubinemia,
hypocalcemia, polycythemia).
- Screening of all pregnant
women should include
- Random (need not be
fasting) venous blood glucose 1 hr after ingestion of 50 gm of glucose at
24-28 wks' gestation. Values >140 mg/dL are indication for 3-hr GTT
with 100 gm glucose. 1-hr, 50-gm test is abnormal in ~15% of pregnant
women, ~14% of whom have abnormal 3-hr OGTT. Sensitivity is ~79%,
specificity is ~87%.
Diagnostic Criteria
- At least two of the
following glucose plasma levels are found on OGTT with 100-gm glucose
loading dose:
- Fasting ≥105
mg/dL
- 1 hr ≥190
mg/dL
- 2 hrs ≥165
mg/dL
- 3 hrs ≥145
mg/dL
- If abnormal results
during pregnancy, repeat GTT at first postpartum visit; if GTT is normal,
diagnose as diabetes mellitus only during pregnancy, but blood glucose
should be tested at every subsequent visit because of increased risk (30%
during next 5-10 yrs) of developing diabetes mellitus. If postpartum GTT
is abnormal, classify as impaired glucose tolerance, impaired fasting
glucose, or diabetes mellitus using above criteria.
- Glycosylated Hb and
fructosamine are not recommended tests for detection of gestational
diabetes.
- For management of
diabetes mellitus during pregnancy, goal is fasting plasma glucose of
60-110 mg/dL and postprandial levels of <150 mg/dL. Measure serum or
24-hr urine estriol for fetal surveillance. Amniotic fluid
lecithin/sphingomyelin ratio, phosphatidylglycerol, shake test, or
fluorescence polarization to evaluate fetal pulmonary maturity.
- During labor, keep
maternal glucose at 80-100 mg/dL; beware of markedly increased insulin
sensitivity in immediate postpartum period.
Laboratory
Evaluation of Fetus
- During third trimester,
urinary estriol level is used as indicator of fetoplacental integrity.
- Placental function is
also indicated by hCG, human placental lactogen, estradiol, and
progesterone levels. Lecithin/sphingomyelin ratio measured on amniotic
fluid is used to predict pulmonary maturity.
- At time of cesarean
section, before opening amniotic sac, obtain sterile sample of amniotic
fluid for culture, Gram stain, lecithin/sphingomyelin ratio.
Diabetes
Mellitus, Neonatal
- Blood glucose is often
between 245 and 2300 mg/dL.
- Metabolic acidosis of
some degree is usually present.
- Ketonuria is variable.
- Laboratory findings due
to dehydration
- Laboratory findings due
to infection or CNS lesions, which are present in one-third of patients
- Has been detected as
early as fourth day. Usually is transient.
- Increased association
with postmaturity, low birth weight, neonatal hypoglycemia, steroid
therapy early in neonatal period.
Laboratory
Evaluation of Infant
- 47% risk of
hypoglycemia Check blood glucose at 1, 2, 3, 6, 12, 24, 36, 48
hrs
- 22% risk of
hypocalcemia Check blood calcium at 6, 12, 24, 48 hrs
- 34% risk of
polycythemia Check Hct at 1, 24 hrs
- 19% risk of
hyperbilirubinemia Check serum bilirubin at 24, 48 hrs
- Sixfold increased risk of
hyaline membrane disease During first hour of life, examine gastric
aspirate with Gram stain for bacteria and PMNs and perform shake test for
lecithin/sphingomyelin
P.620
|
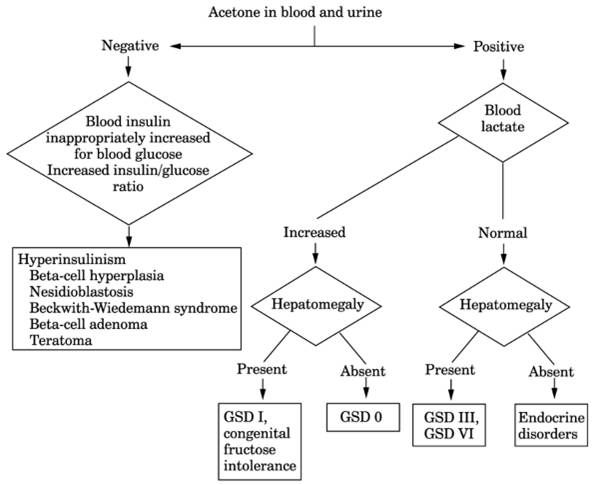
|
|
Fig. 13-9. Algorithm for neonatal
hypoglycemia. (GSD = glycogen storage disease.)
|
- 9% risk of major
congenital anomalies (e.g., cardiac, renal) and other problems (e.g.,
renal vein thrombosis, excess mucus) Check blood gases for
evaluation of ear, nose, and throat, umbilicus, rectum, urine, blood, CSF,
gastric aspirate
Glucagonoma
- (Arise
from alpha cells of pancreatic islets. 60% are malignant.)
- Diabetes mellitus
- Anemia
- Increased serum insulin level is characteristic
- Increased serum level of glucagon. Serum proglucagon is also increased occasionally.
- ○ Clinical clue is
association of dermatitis (necrolytic migratory erythema) with
insulin-requiring diabetes.
Hyperglycemia,
With Heterogeneous Genetic Diseases
- Alström's syndrome
- Ataxia-telangiectasia
- Diabetes mellitus
- Friedreich's ataxia
- Hemochromatosis
- Herrmann's syndrome
- Hyperlipoproteinemias
(three different types)
- Isolated growth hormone
(GH) deficiency
- Laurence-Moon-Bardet-Biedl
syndrome
- Lipoatrophic diabetes
- Myotonic dystrophy
- Optic atrophy
P.621
- Prader-Willi syndrome
- Refsum's syndrome
- Schmidt's syndrome
- Werner's syndrome
Hypoglycemia,
Classification
- See Table
13-13.
- Diagnosis requires triad of low blood glucose at the time of spontaneous hypoglycemic
symptoms and alleviation by administration of glucose that corrects
hypoglycemia. (Glucose concentration is 15% lower in
whole blood than in serum or plasma.)
- Reactive (i.e., after
eating)
- Alimentary (rapid
gastric emptying, e.g., after subtotal gastrectomy, vagotomy)
- Impaired glucose
tolerance as in diabetes mellitus (mild maturity onset)
- Functional (idiopathic)
- Rare conditions (e.g.,
hereditary fructose intolerance, galactosemia, familial fructose and
galactose intolerance)
- Fasting
(spontaneous)-almost always indicates organic disease
- Liver-severe parenchymal
disease (including sepsis, congestive heart failure, Reye's syndrome) or
enzyme defect (e.g., glycogen storage diseases, galactosemia)
- Chronic renal
insufficiency
- Pancreatic
- Insulinoma (pancreatic
islet cell tumor)
- MEN type I
- Pancreatic hyperplasia
- Deficiency of hormones
that oppose insulin (e.g., decreased function of thyroid, anterior
pituitary, or adrenal cortex)
- Postoperative removal of
pheochromocytoma
- Large extrapancreatic
tumors (65% are intra- or retroperitoneal fibromas or sarcomas)
- Certain epithelial
tumors (e.g., hepatoma, carcinoid, Wilms' tumor)
- Drugs (including
factitious use)
- Insulin
- Sulfonylureas
- Alcohol
- Salicylates
- Pentamidine
- Quinine
- Propranolol (rare)
- Others may potentiate
effect of sulfonylurea (e.g., sulfonamides, butazones, coumarins,
clofibrate)
- Artifactual (high WBC or
RBC count, e.g., leukemia or polycythemia)
- Starvation, anorexia
nervosa, lactic acidosis, intense exercise
- Insulin antibodies or
insulin receptor antibodies
- Combined reactive and
fasting types
- Insulinoma
- Adrenal insufficiency
- Insulin antibodies or
insulin receptor antibodies
Infants
- See Fig.
13-9.
- Transient
(<14 days)
- Symptomatic or
asymptomatic; occurs in 1-3 in 1000 full-term infants
- Maternal (e.g.,
diabetes, toxemia, complicated labor or delivery)
- Infant, e.g.,
- Prematurity, small size
for gestational age
- Intrauterine
malnutrition
- Erythroblastosis
- Secondary, e.g.,
sepsis, asphyxia, anoxia, cerebral or subdural hemorrhage
- Congenital anomalies
- Iatrogenic, e.g.,
postoperative complications, abrupt cessation of glucose infusion, after
exchange transfusion, cold injury
P.622
|
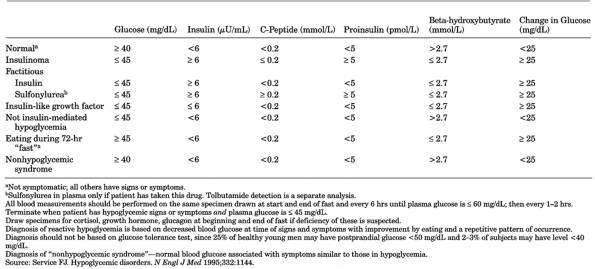
|
|
Table 13-13. Laboratory Interpretation of
72-Hr Fast for Hypoglycemia
|
P.623
- Persistent
- Hyperinsulinism
- Beta cell hyperplasia
- Nesidioblastosis
- Beckwith-Wiedemann
syndrome (children also have Wilms' and other embryonal tumors,
visceromegaly, macroglossia)
- Beta cell tumor
- Teratoma
- Endocrine disorder
- Hypothyroidism
- Congenital adrenal
hyperplasia (CAH)
- Anterior pituitary
hypofunction
- Decreased glucagon
- Hepatic enzyme
deficiencies
- Glycogen storage
disease types I, III, VI, 0
- Congenital fructose
intolerance
- Galactose 1-phosphate
deficiency
- Maple syrup urine
disease
- Galactosemia
- Hereditary tyrosinemia
- Methylmalonic acidemia
- Propionicacidemia
Hypoglycemia,
Factitious
- See Table
13-13, and Fig. 13-10.
Due to
Insulin
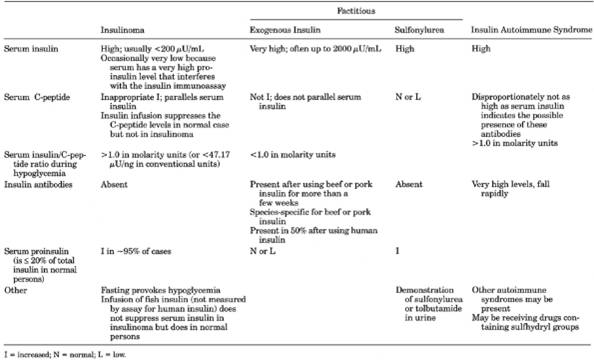
- During hypoglycemic episode, high insulin and low C-peptide levels in serum confirm
diagnosis of exogenous insulin administration (diagnostic triad).
(Increased endogenous insulin secretion is always associated with
increased secretion of C-peptide, which is the part of the proinsulin
molecule cleaved off when insulin is secreted and therefore is produced in
equimolar amounts with insulin.)
- Insulin/glucose ratio
>0.3 in serum (normal is <0.3). Increased ratio is also seen in
autonomous production due to insulinoma.
- Extreme elevations of serum insulin (e.g., >1000 µU/mL) suggest
factitious hypoglycemia (fasting levels in patients with insulinoma are
rarely >200 µU/mL).
- Insulin antibodies appear
in 90% of persons injected with beef or pork insulin and 50% of those injected
with human insulin for more than a few weeks but are almost never present
in persons not taking insulin (rarely occur on an autoimmune basis),
although this indicator may be less useful with the future use of more
purified and human insulin.
Due to
Sulfonylureas
- Biochemically
indistinguishable from insulinoma (increased serum C-peptide and insulin
levels with insulin/C-peptide molar ratio <1.0).
- Specific chemical assay can identify the agent in serum or urine.
Due to
Tolbutamide
Acidification of urine causes a white precipitate due to formation of
carboxytolbutamide.
Hypoglycemia,
Leucine-Induced
- Symptoms within 30 mins
of consumption of high-protein meal or after prolonged fast. May be
neonatal or appear later in first year; symptoms become increasingly
severe; tends to improve spontaneously by 4-6 yrs of age.
- Blood glucose falls
>50% within 20-45 mins after oral administration of L-leucine. Same result in 70% of patients with insulinoma.
P.624
|
|
|
Table 13-14. Comparison of Laboratory
Findings in Causes of Hypoglycemia
|
P.625
|
|
|
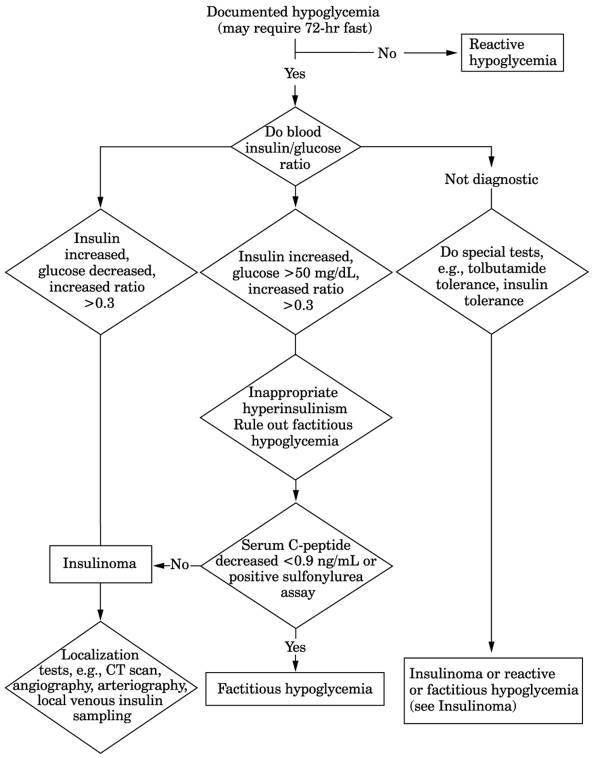
Fig. 13-10. Algorithm for diagnosis of
suspected insulinoma. (CT = computed tomography.)
|
Hypoglycemia,
Neonatal
Diagnostic
Criteria
- Plasma glucose <25
mg/dL in low-birth-weight infants.
- Plasma glucose <35
mg/dL in normal-birth-weight infants in first 72 hrs. Plasma glucose
<45 mg/dL in normal-birth-weight infants after 72 hrs.
- Make diagnosis on basis
of two abnormal glucose values, e.g., two plasma levels, one plasma and
one CSF level.
- Plasma glucose values
are 14% higher than whole blood values.
- Capillary blood samples
should be taken from warm heel and transported to laboratory on ice (at
room temperature, blood glucose level decreases by 18 mg/dL/hr).
- Determination by
measurement of reducing substances may give falsely elevated levels for
glucose because in blood of newborns, non-glucose-reducing substances
P.626
may range up to 60 mg/dL. Therefore use techniques that measure glucose
specifically, e.g., glucose oxidase.
- Bedside glucose monitor
utilizing dipsticks should not be used for this diagnosis-is not reliable
in the low glucose range.
Due To
- Transient symptomatic or
asymptomatic hypoglycemia may be associated with delayed feeding, toxemia,
perinatal asphyxia, twin birth, hypothermia, or low birth weight, or may
be idiopathic. Occurs in 1-3 in 1000 full-term infants.
- Hyperinsulinism (e.g.,
maternal diabetes, erythroblastosis, Beckwith-Wiedemann syndrome,
insulinoma, maternal drug therapy or starvation)
- Hormone deficiencies (e.g.,
hypothyroidism, pituitary hypofunction, adrenal insufficiency or
unresponsiveness)
- Hereditary metabolic
disorders
- Galactosemia
- Type I glycogen storage
disease
- Amino acid disorders
(e.g., tyrosinemia type I)
- Organic acid disorders
(e.g., methylmalonicacidemia, propionicacidemia)
- Carnitine deficiency
disorders (e.g., carnitine palmityl transferase deficiency)
- Disorders of fat
oxidation (e.g., medium-chain acyl-CoA dehydrogenase deficiency)
- Disorders of
gluconeogenesis (e.g., pyruvate carboxylase deficiency)
- Others
- Iatrogenic (e.g., after
exchange transfusion)
- Miscellaneous (e.g.,
sepsis diarrheal illness, CNS abnormalities, congenital heart disease)
Infants
of Diabetic Mothers
- Blood glucose is <30
mg/dL in ≤50% of infants of diabetic mothers; usually asymptomatic;
usually within first hours after birth. Hypocalcemia is common in these
cases and occurs at 24-36 hrs after birth.
- Glucose levels should be
checked every hour for first 6 hrs of life.
Insulin
Autoimmune Syndrome
- (Cause
is unknown but patients may be receiving drugs containing a sulfhydryl
group [e.g., pyritinol]. Associated with other autoimmune syndromes [e.g.,
hyperthyroidism, autoimmune thrombocytopenia with primary biliary
cirrhosis]. Appears to be a self-limiting condition.)
- Fasting hypoglycemia.
- ○ Elevated serum
insulin and C-peptide levels that are discordant (insulin/C-peptide molar
ratio >1.0) indicates the possible presence of these antibodies. These
elevations are artifactual due to effect of antibody on assay method.
- May be difficult to distinguish
from factitious hypoglycemia.
- Extremely high levels of antiinsulin antibodies that rapidly decrease.
- Never any prior exposure
to exogenous insulin.